Pfizer data suggest third dose of Covid-19 vaccine 'strongly' boosts protection against Delta variant | CNN

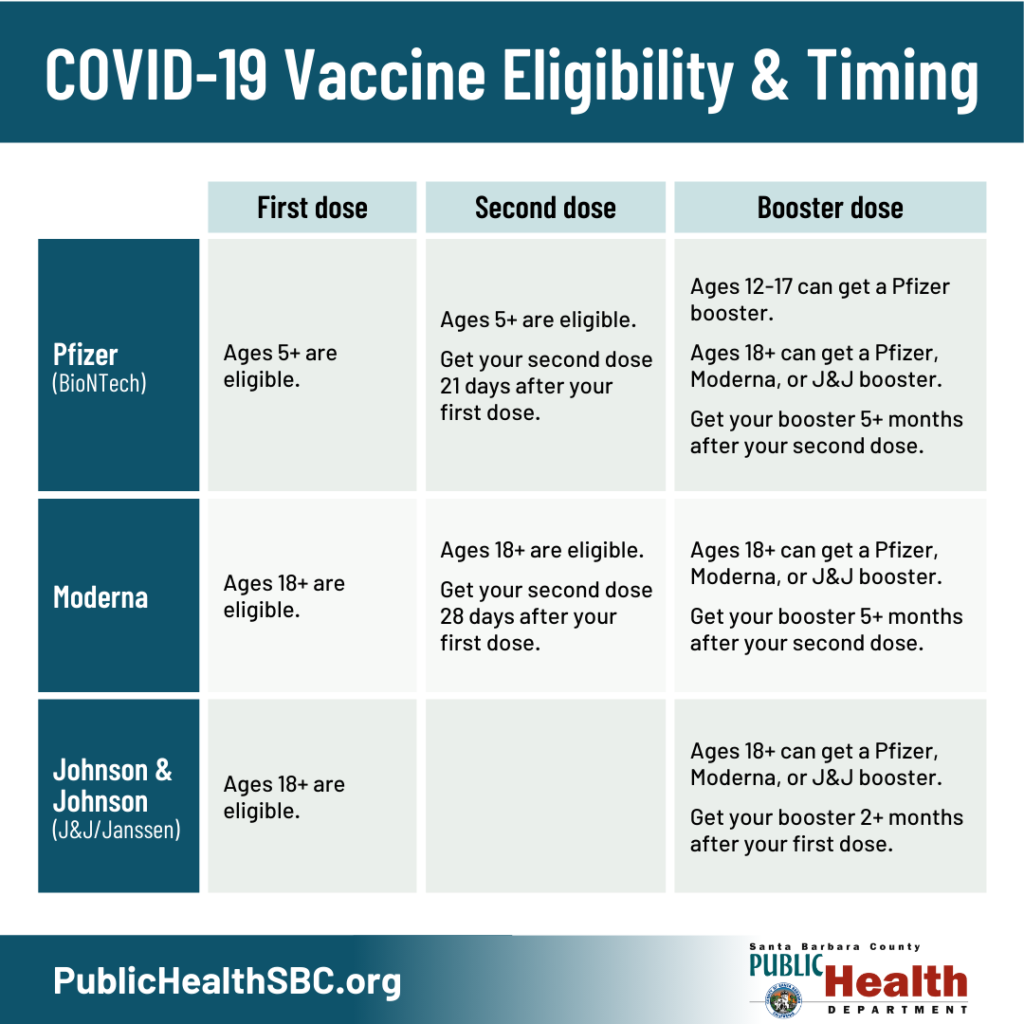
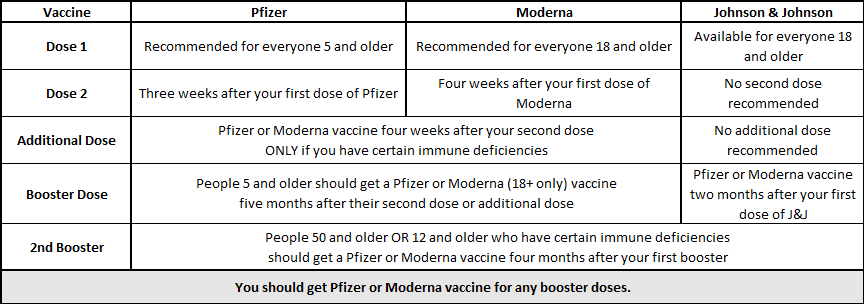
FDA, CDC, Western States authorize booster doses of the Pfizer COVID-19 vaccine for 16- and 17-year-olds : Oregon Health News Blog

Frederick County Health Department - Are you eligible for a Pfizer booster dose of COVID-19? Yes if you got Pfizer doses 1 & 2, your 2nd dose was more than 6 months

Covid-19, Fda estende l'idoneità per la dose booster del vaccino Pfizer-BioNTech ai bambini da 5 a 11 anni - AboutPharma

Pfizer-BioNTech booster for specific groups recommended by Western States Scientific Safety Review Workgroup : Oregon Health News Blog
Pfizer, BioNTech say bivalent COVID-19 booster shot performs better against BA.5 omicron subvariant - ABC News